I led the redesign of SOPHiA DDM, SOPHiA Genetics’ flagship SaaS platform for detecting genetic alterations and delivering clinical insights. This initiative was a cornerstone of a significant technical transformation, transitioning the application from a legacy desktop environment to a modern web-based platform. In addition to overseeing the technical migration, my primary goal was to elevate the user experience by introducing advanced UX/UI standards, streamlining workflows for clinical users, and ensuring the platform remained compliant with regulatory and healthcare industry requirements.
Challenge
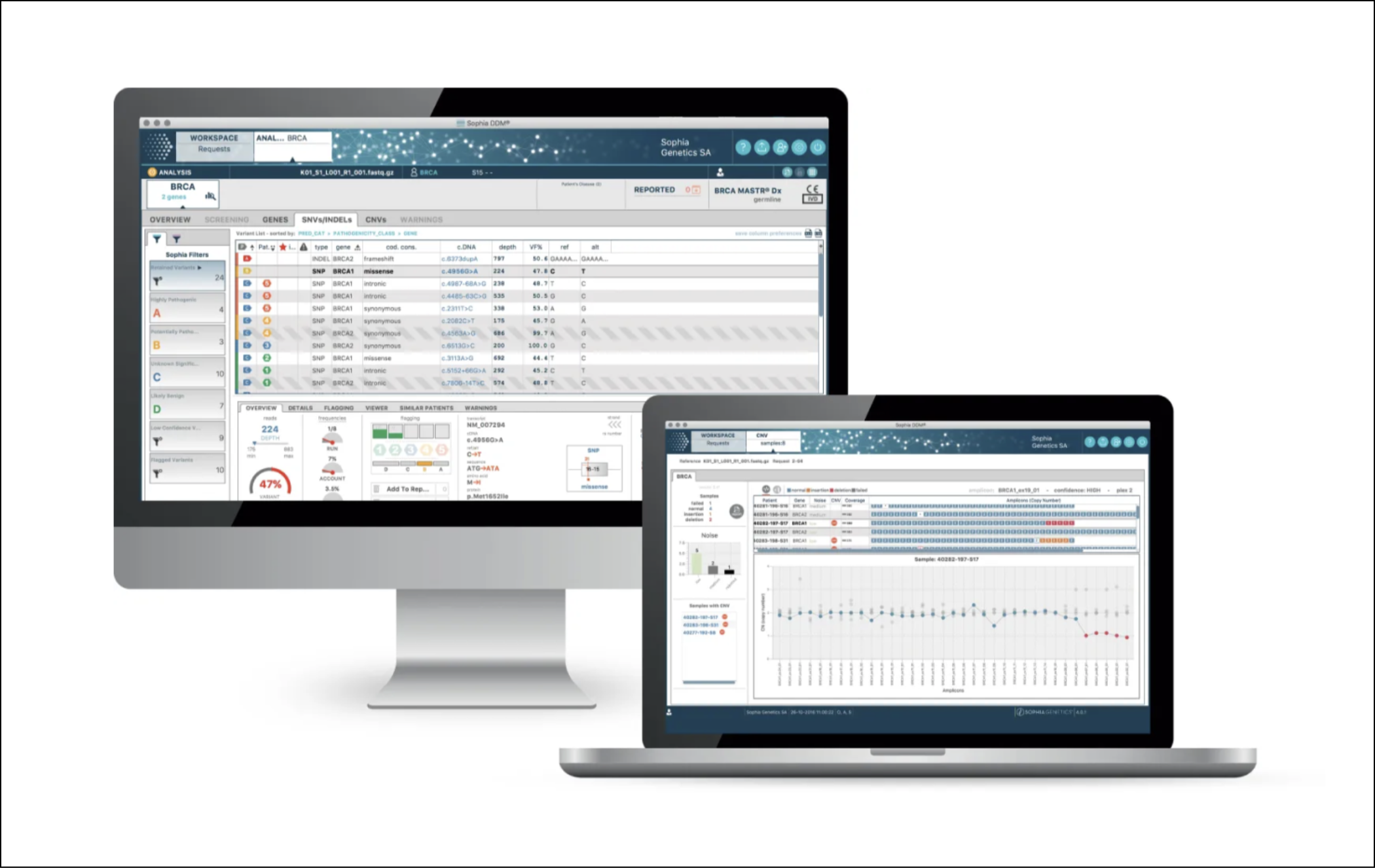
Over more than 11 years of evolution, the platform had grown increasingly complex and dense, shaped by legacy technical constraints and incremental feature additions. Redesigning such a system demanded a careful balance between innovation and continuity, as clinical users depended on familiar workflows in a high-stakes environment where reliability was paramount. Key challenges included a tight two-year development timeline, the need to present complex genomic data with clarity and precision, and the need to support a diverse set of user profiles with varying goals and expertise. Additionally, portions of the platform had to remain compliant with stringent regulatory constraints and documentation requirements for IVD marking, making every design decision subject to both usability and rigorous traceability and validation standards.
Solution
Created a comprehensive design system with 50+ reusable components, detailed documentation, and clear integration guidelines. Established governance processes to ensure the system evolves with product needs while maintaining consistency.
User Research
We conducted in-depth interviews with 15 customers to uncover pain points, challenge assumptions, and prioritize actionable improvements.
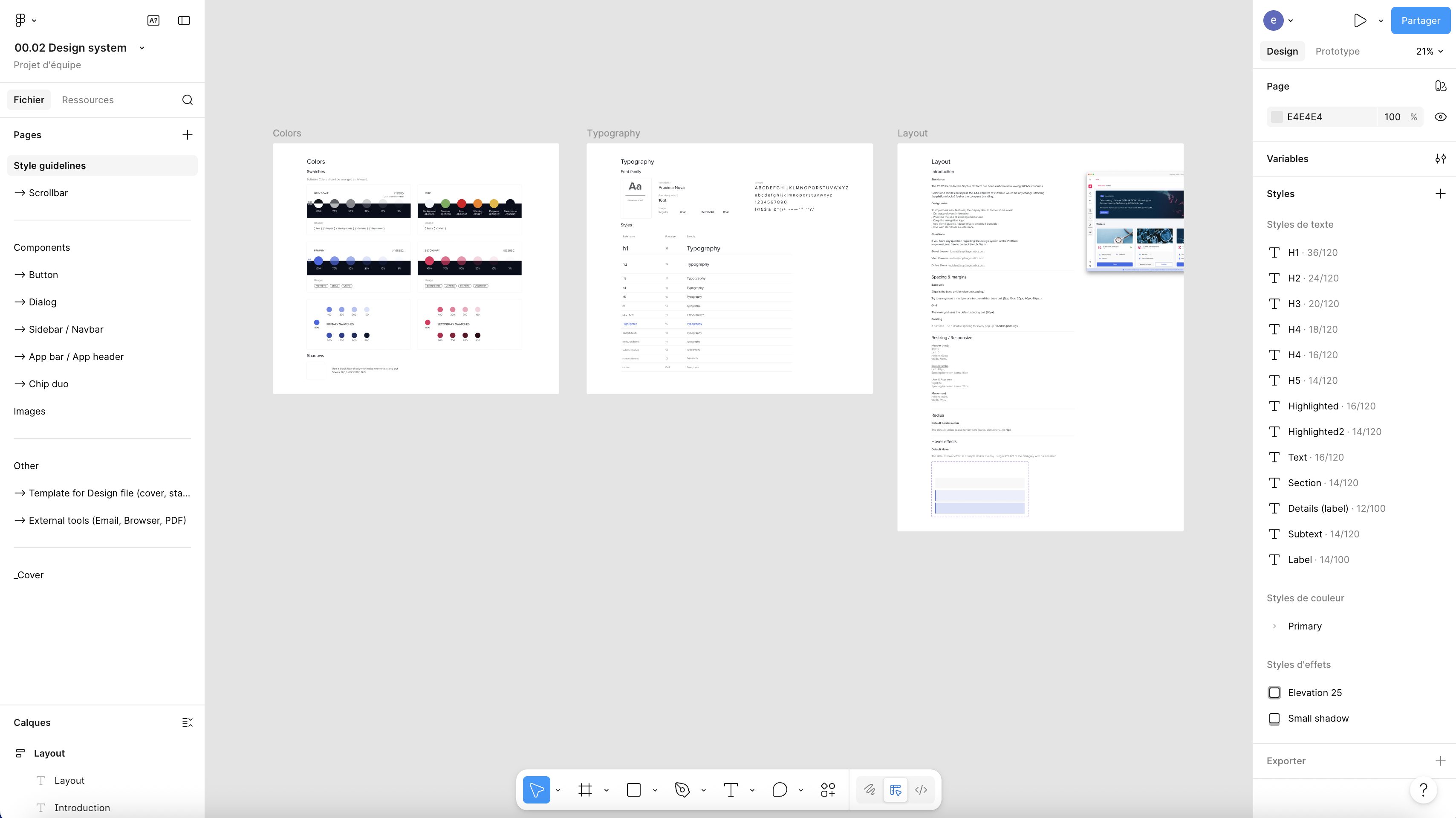
Design System
Leveraging these insights, I defined the new UX direction and spearheaded the creation of the company's first comprehensive design system, combining Material UI X components with 50+ custom reusable components to establish scalable interaction patterns and visual standards. I worked hand in hand with development teams to translate this design system into an SDK, ensuring consistency and long-term scalability across the platform.
Content Strategy
In parallel, I developed a robust content strategy, encompassing style guides, terminology standards, and localization workflows,to drive clarity, consistency, and global accessibility. I also directed the development of a modern, accessible visual design aligned with healthcare standards and compliance expectations.
Implementation and Validation
Throughout the project, I led the design validation process by building interactive prototypes and conducting usability testing sessions with clinical users to confirm design decisions and refine workflows. I partnered closely with engineering teams to ensure design fidelity, supported a phased rollout strategy to minimize disruption for existing customers, and meticulously documented design specifications and decisions to meet IVD marking requirements.
Result and Impact
The redesigned platform launched successfully in September 2024, delivering a modern web-based experience while preserving essential clinical workflows. The new interface improved usability, consistency, and scalability by introducing a robust design system and standardized UX patterns. The project also fostered stronger collaboration between product, design, and engineering through the SDK approach and structured design processes.
Before/After
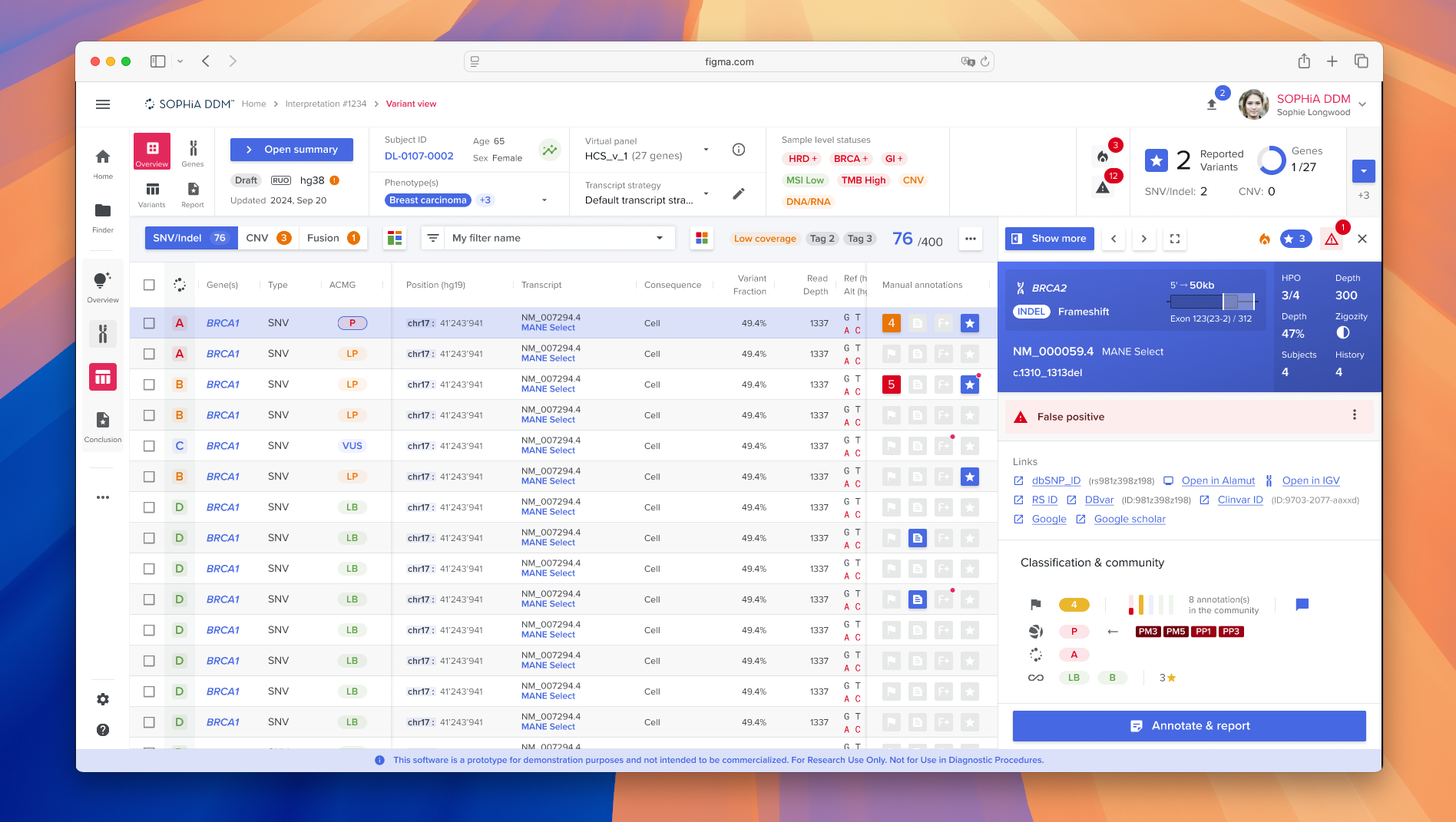
The Platform in Action
Design System